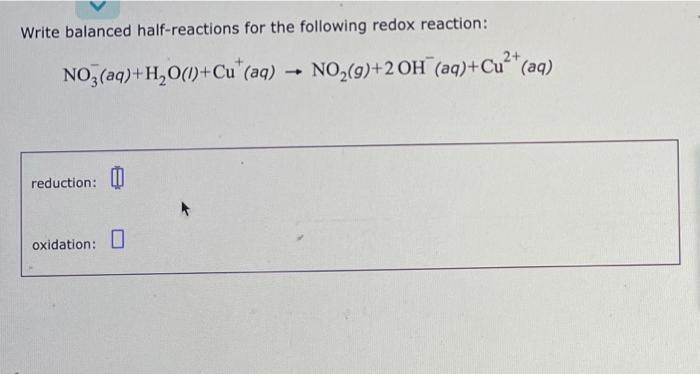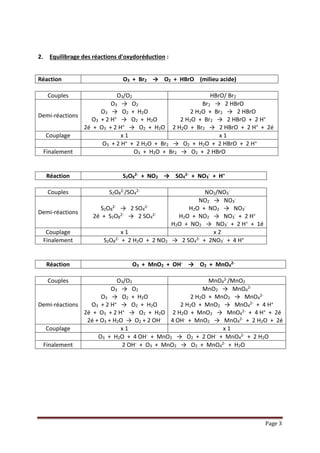
Solar Splitting of H2O and CO2 via Thermochemical Redox Cycles – Professorship of Renewable Energy Carriers | ETH Zurich

SOLVED: The net redox reaction in a fuel cell is given below: 2H2 + O2 -> 2H2O What is the reaction at the anode in a fuel cell? O2 + 4e- ->
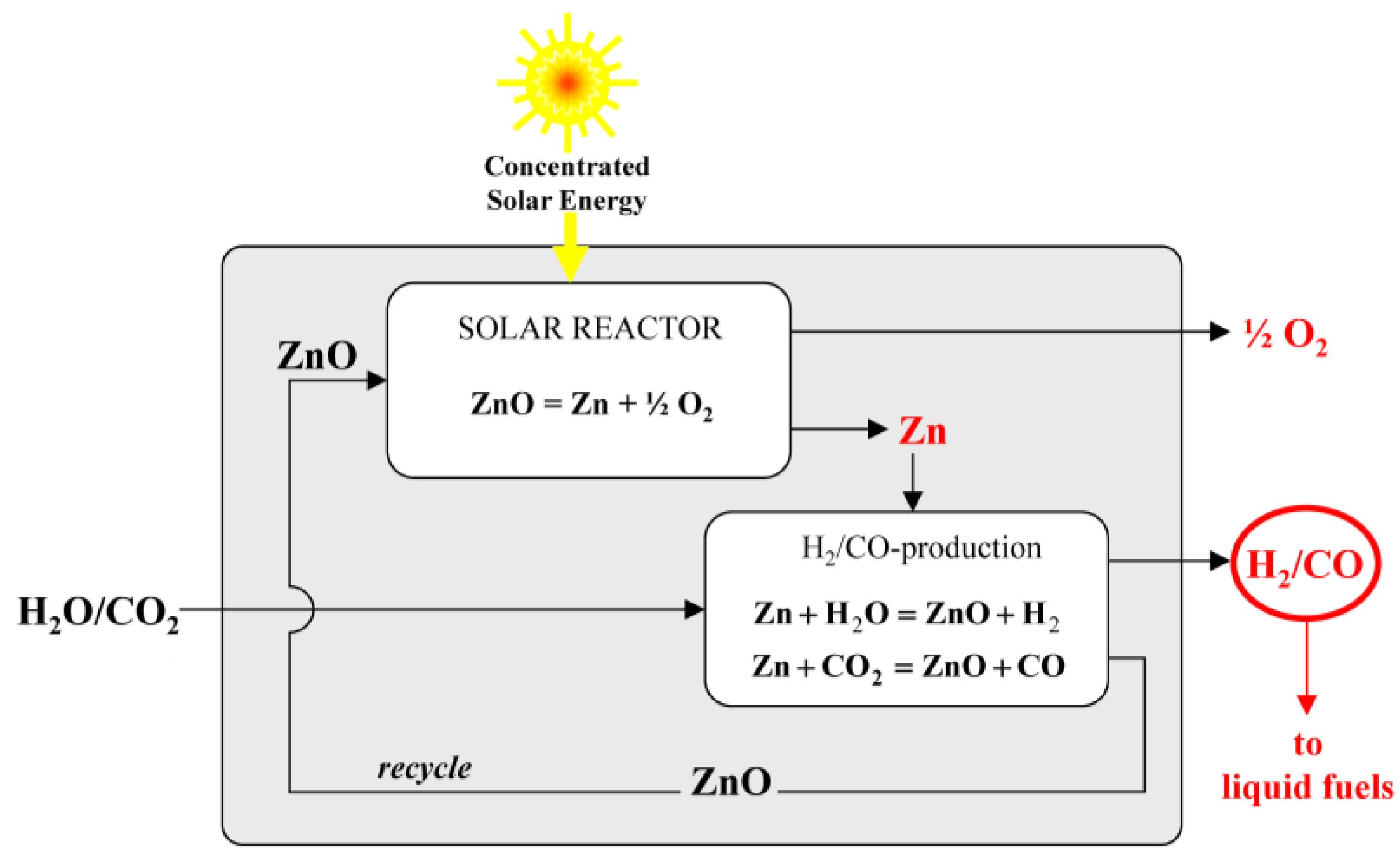
Materials | Free Full-Text | Review of the Two-Step H2O/CO2-Splitting Solar Thermochemical Cycle Based on Zn/ZnO Redox Reactions

SOLVED: Balance the following redox reaction in basic aqueous solution: Mn2+ + H2O2 -> MnO2 + H2O a. Mn2+ + H2O2 -> MnO2 + 2 OH b. 2 OH- + Mn2+ +

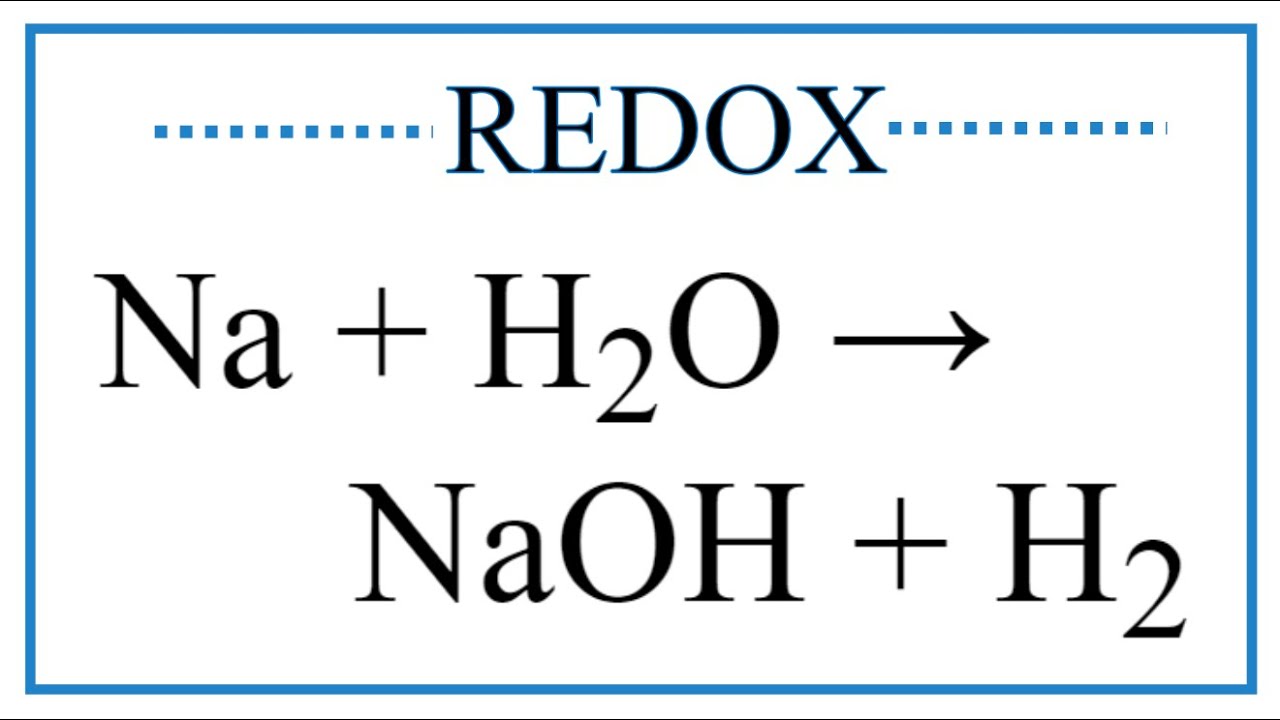
2. Which of the following is not a redox reaction? (a) CuO + H2 → Cu + H2O (b) Na + H2O → NaOH + H, (c) CaCO3 → CaO + CO2 (d) 2K+F2 → 2KF

High-temperature heat recovery from a solar reactor for the thermochemical redox splitting of H2O and CO2 – HeliosCSP – Portal de noticias de energía termosolar