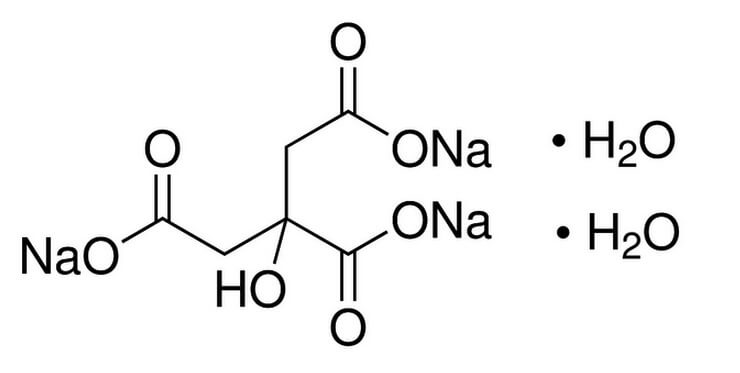
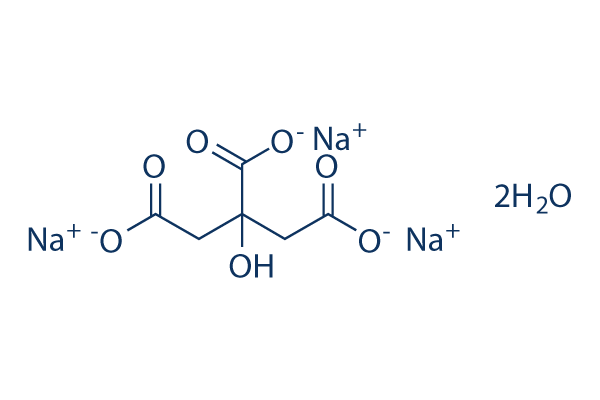
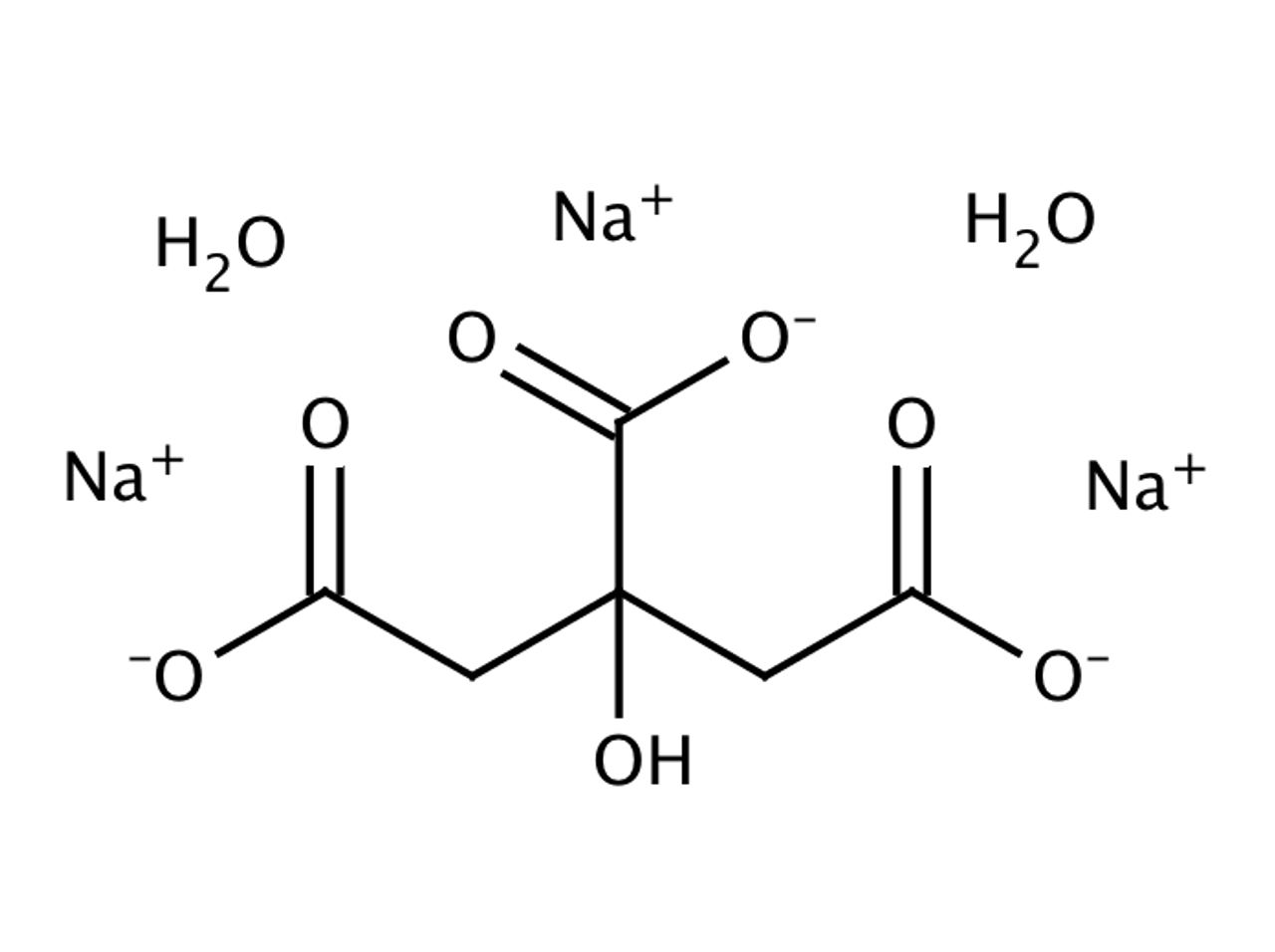
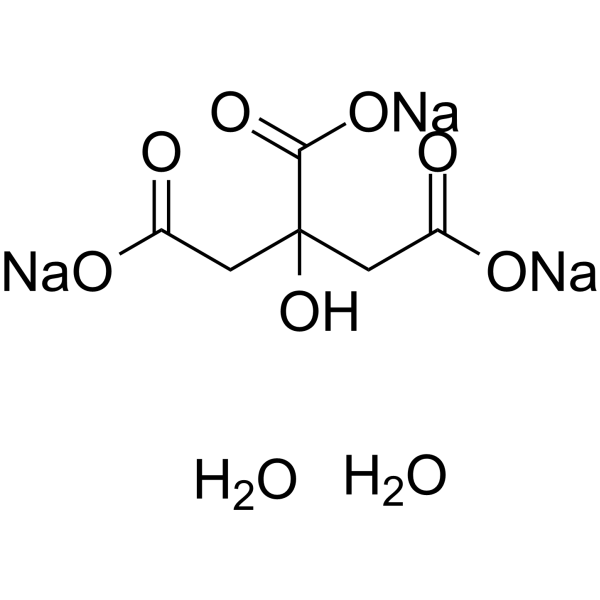
SOLVED: Alka-Seltzer contains sodium bicarbonate (NaHCO3) and citric acid (H3C6H5O7). When the tablet is dissolved in water, the following reaction produces sodium citrate, water, and carbon dioxide: 2NaHCO3 + H3C6H5O7 â†' Na2C6H5O7 +
![Phase diagrams for [C4mim]BF4 + Na3C6H5O7 + H2O ABS in the presence of... | Download Scientific Diagram Phase diagrams for [C4mim]BF4 + Na3C6H5O7 + H2O ABS in the presence of... | Download Scientific Diagram](https://www.researchgate.net/publication/325860685/figure/fig1/AS:961857821081639@1606336183966/Phase-diagrams-for-the-C4mimBF4-Na3C6H5O7-H2O-ABS-in-the-presence-of-amino-acids-of_Q320.jpg)
Phase diagrams for [C4mim]BF4 + Na3C6H5O7 + H2O ABS in the presence of... | Download Scientific Diagram

Reduction of HAuCl4 with sodium citrate in water: the sizes of AuNPs... | Download Scientific Diagram
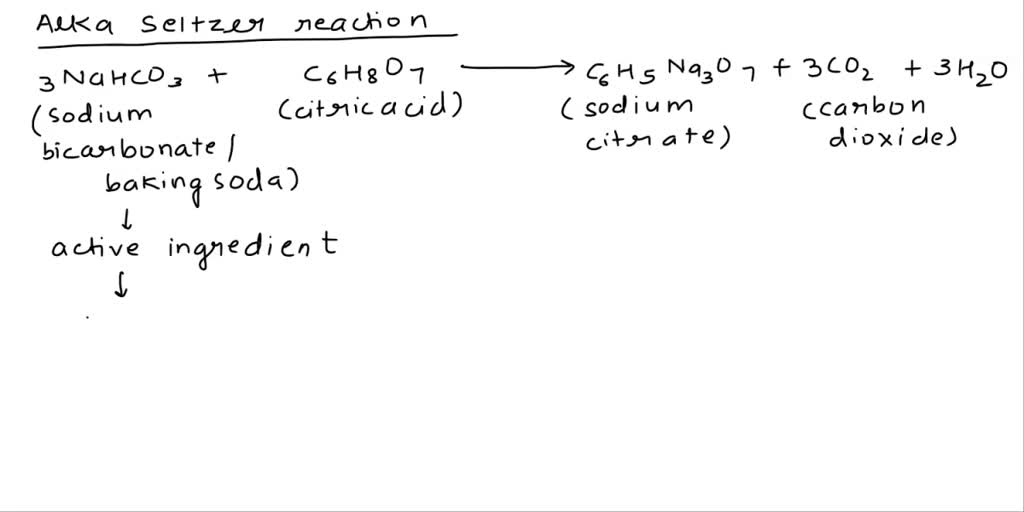
SOLVED: Classify each component of the reaction that occurs when an Alka-Seltzer tablet is dissolving in water: NaHCO3, C6H8O3, H2O, CO2, Na3C6H5O7.
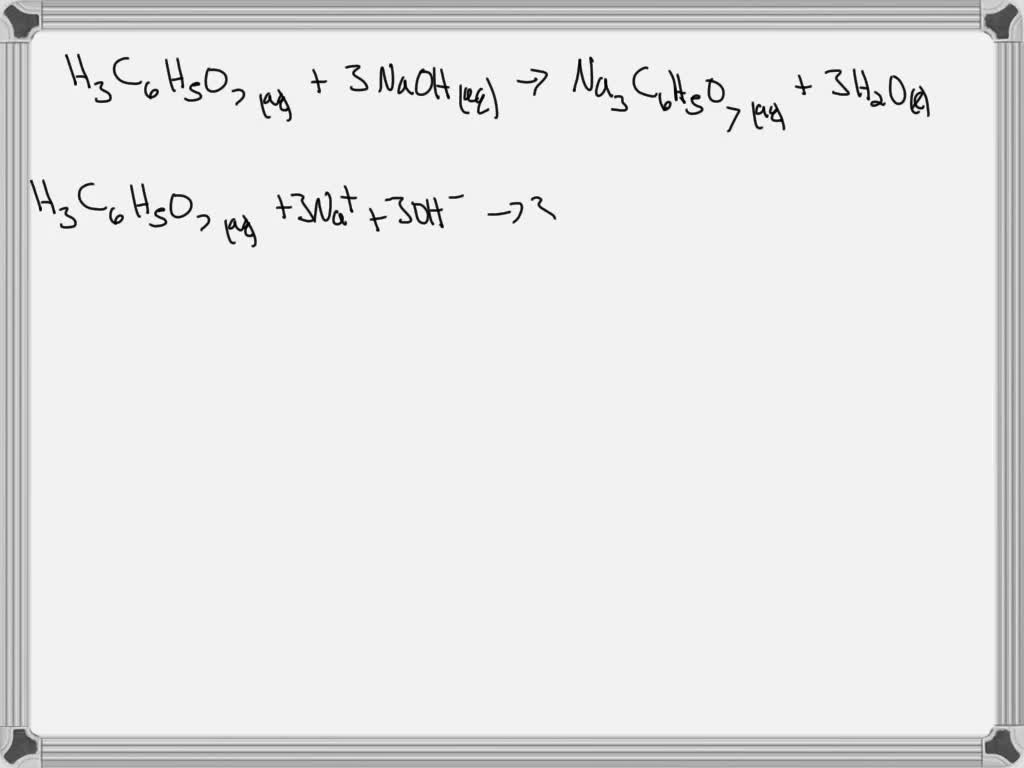
SOLVED: H3C6H5O7(aq) + 3 NaOH(aq) = Na3C6H5O7(aq) + 3 H2O(l) write complete ionic and net-ionic equations

How to Balance H3C6H5O7 + NaHCO3 = CO2 + H2O + Na3C6H5O7 (Citric acid + Sodium bicarbonate ) - YouTube
![Phase diagrams for [C4mim]BF4 + Na3C6H5O7 + H2O ABS in the presence of... | Download Scientific Diagram Phase diagrams for [C4mim]BF4 + Na3C6H5O7 + H2O ABS in the presence of... | Download Scientific Diagram](https://www.researchgate.net/publication/325860685/figure/fig4/AS:961857825292304@1606336184782/Phase-diagrams-for-C4mimBF4-Na3C6H5O7-H2O-ABS-in-the-presence-of-different-mass.png)
Phase diagrams for [C4mim]BF4 + Na3C6H5O7 + H2O ABS in the presence of... | Download Scientific Diagram

SOLVED: The Alka Seltzer reaction involves citric acid and sodium bicarbonate (baking soda) according to the following unbalanced equation: H3C6H5O7 + NaHCO3 -> Na3C6H5O7 + H2O + CO2. Starting from 1.3 g

SOLVED: Aka-setter contains sodium bicarbonate (NaHCO3) and the citric acid (H3C6H5O7). When the tablet is dissolved in water, the following reaction produces sodium citrate (Na3C6H5O7), water (H2O), and carbon dioxide gas (CO2): (