A 0.492 g of KH_2PO_4 is titrated against a solution of 0.112 M NaOH. The volume of the base required to do this is 25.6 mL. The reaction involved is, KH_2PO_4 +

Thermodynamic Properties Data of Ternary System KBr–KH2PO4–H2O at 298.15 K | Journal of Chemical & Engineering Data
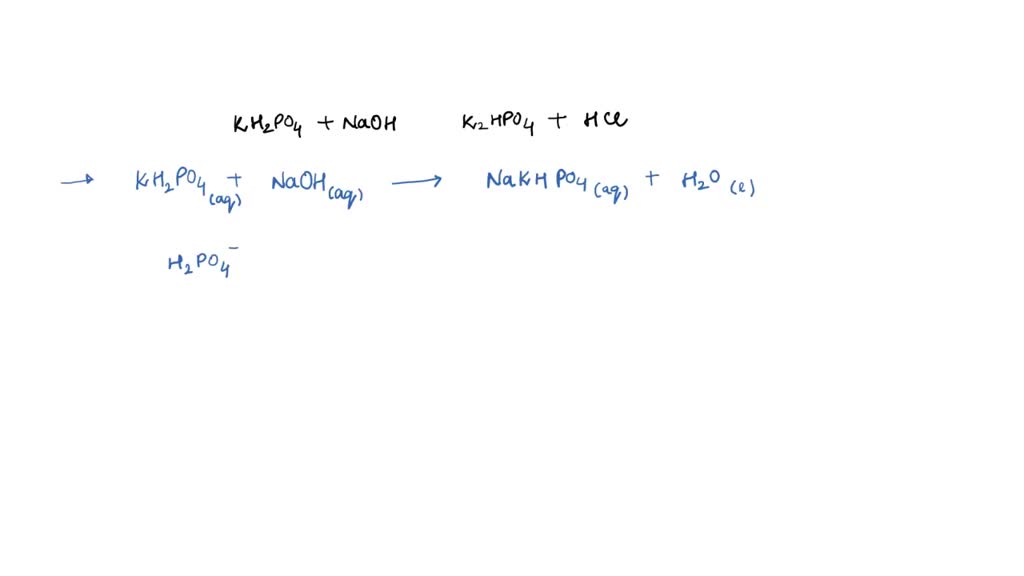
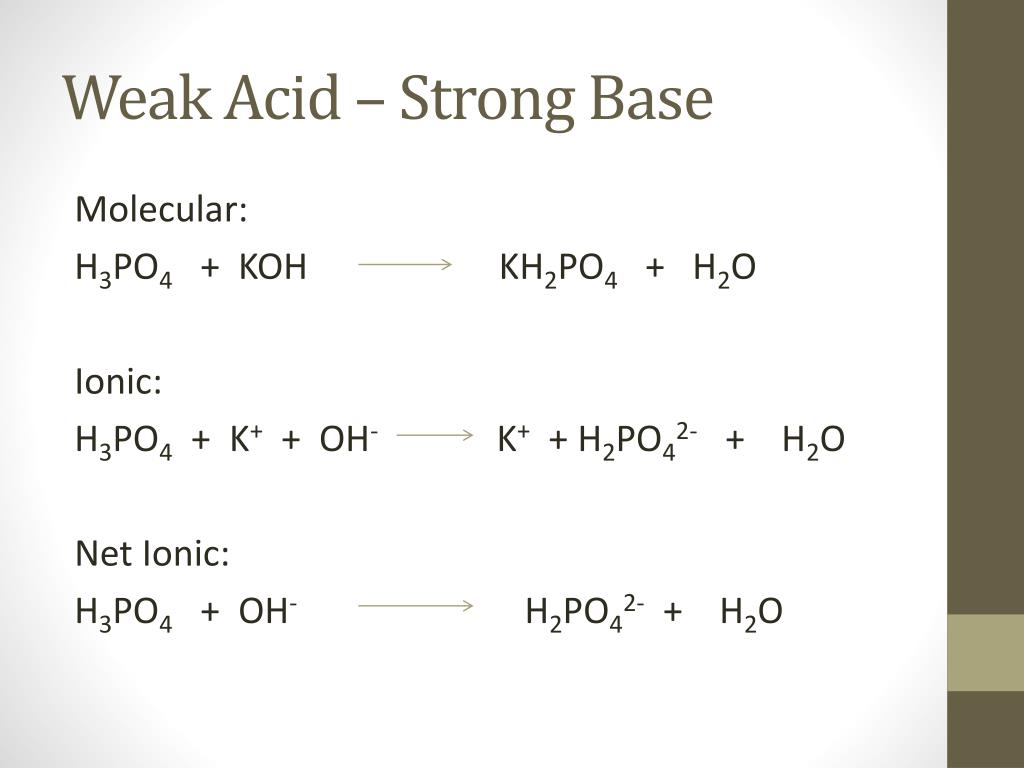
SOLVED: Write the equation and the reaction of the buffer solution KH2PO4 /K2HPO4 when NaOH and HCl is added

SOLVED: A 0.492-g sample of KH2PO4 is titrated with 0.112 M NaOH, requiring 25.6 mL: H2PO4- + OH- → HPO42- + H2O What is the percent purity of the KH2PO4 (FW = 136.09)?

OneClass: Spectrophotometric analysis of phosphate can be performed by the following procedure: A. KH...

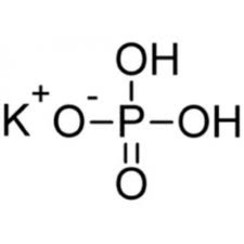
SRL Potassium Dihydrogen Orthophosphate for molecular biology, 99.5% 500Gm, CAS 7778-77-0, Molecular Formula : KH2PO4, Storage : Room Temperature, Shelf Life : 60 Months for laboratory use only : Amazon.in: Industrial & Scientific

Calculate the pH of a buffer solution obtained by dissolving 25.0 g of KH2PO4(s) and 38.0 g of Na2HPO4(s) in water and then diluting to 1.00 L. | Homework.Study.com

KH2PO4 crystallisation from potassium chloride and ammonium dihydrogen phosphate – topic of research paper in Chemical sciences. Download scholarly article PDF and read for free on CyberLeninka open science hub.



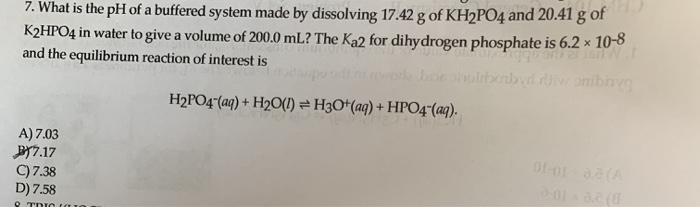
![ANSWERED] c 3 136 A 0 5 g sample of KH PO4 is titra... - Physical Chemistry - Kunduz ANSWERED] c 3 136 A 0 5 g sample of KH PO4 is titra... - Physical Chemistry - Kunduz](https://media.kunduz.com/media/sug-question-candidate/20200703122613132777-1655732.jpg)