CIRAS-4 Portable CO2/H2O Gas Analysis System, Standard system including main console, dual CO2 & H2O IRGAs, air supply unit, user interface, custom transport case, operation manual and basic spares kit. | John

Contrôle de la fenêtre d'un analyseur rapide CO2/H2O placé sur une "tour à flux" destinée à mesurer | CNRS Images

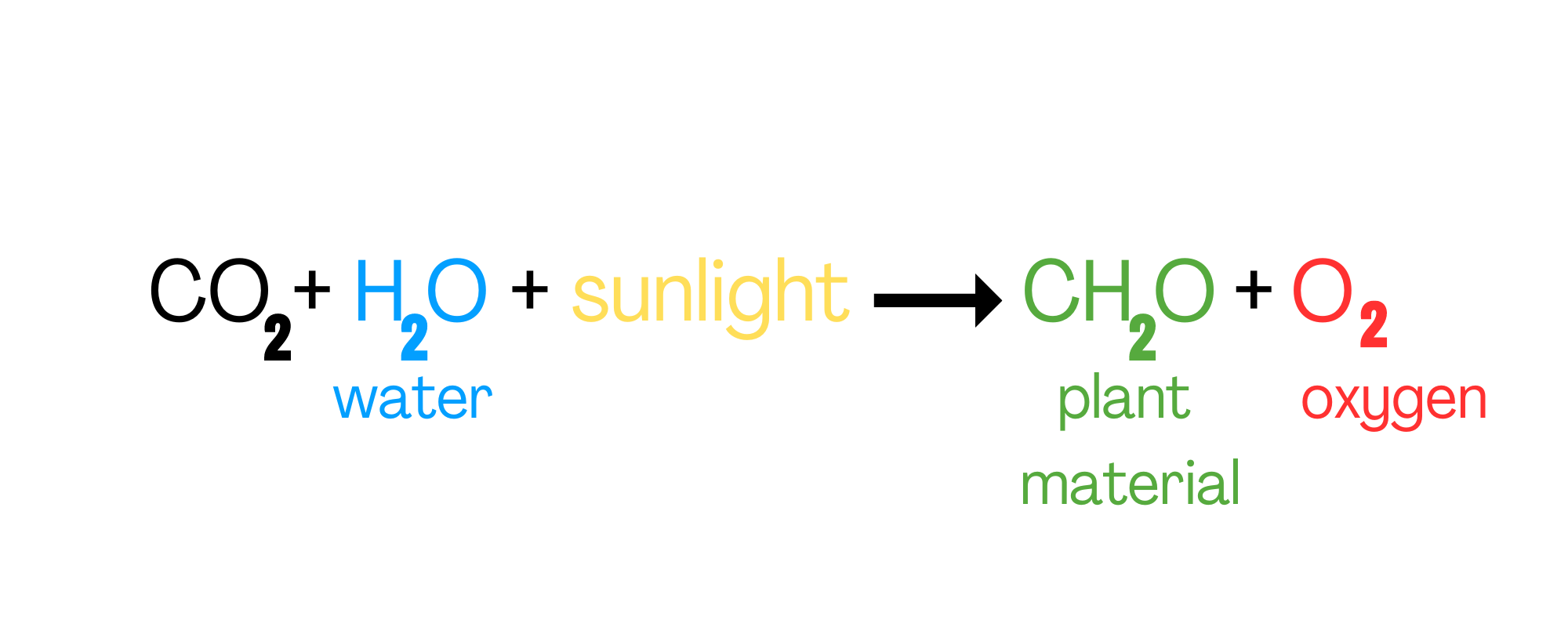
CO2<=>H2O Global Warming According to NASA : ...WaterVapor=H20=62% of the greenhouse effect ...CarbonDioxide=CO2=22% of the greenhouse effect
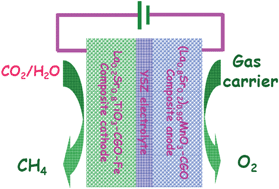
Direct synthesis of methane from CO2/H2O in an oxygen-ion conducting solid oxide electrolyser - Energy & Environmental Science (RSC Publishing)
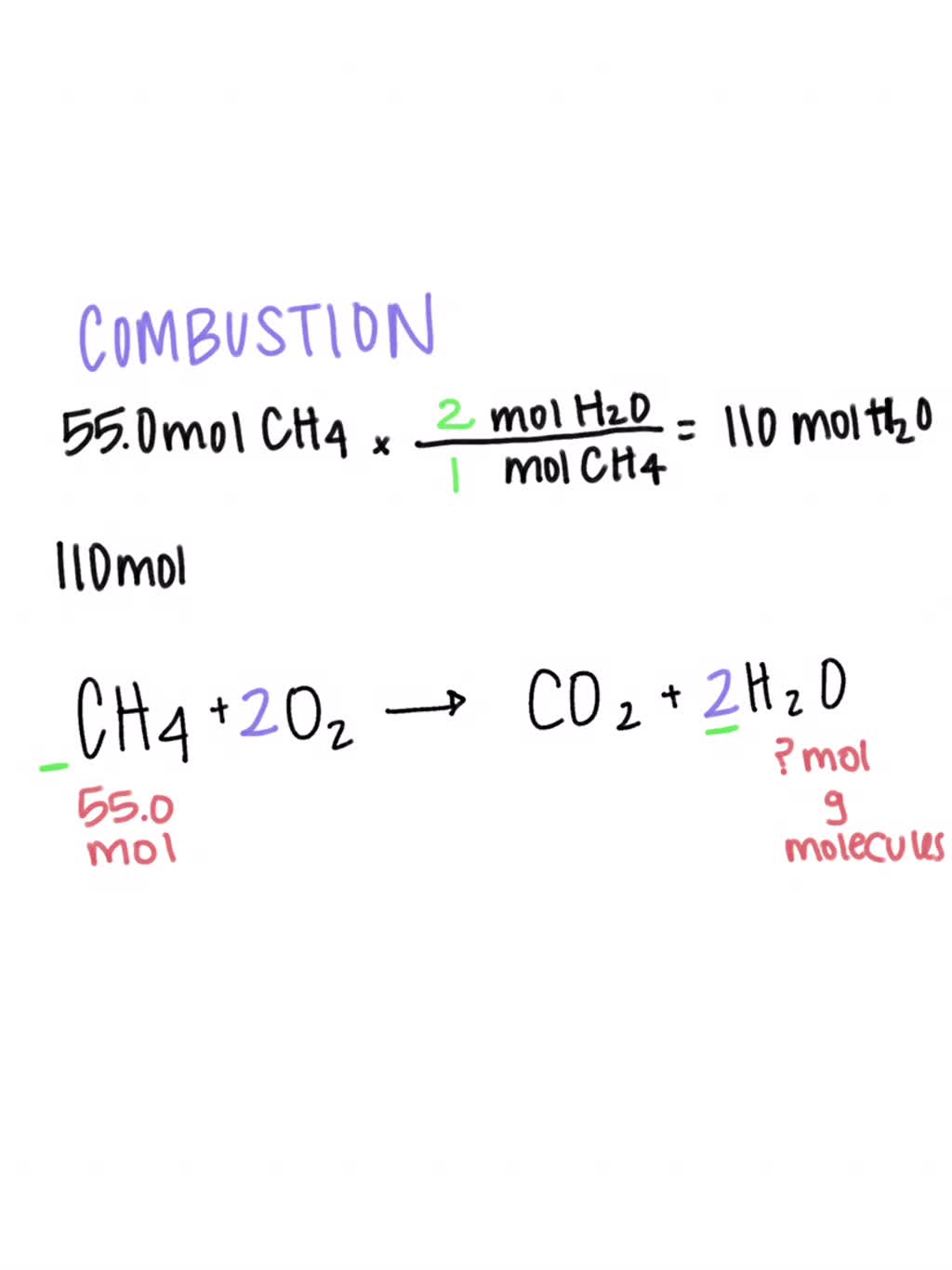
SOLVED: CH4 + O2 ——————- CO2 + H2O What type of reaction does this equation represent? Write and balance the equation. Assume that there are 55.0 moles of CH4. How many moles
Write fully balanced equations for the following : (a) CO2 + H2O → ............ - Sarthaks eConnect | Largest Online Education Community

of breakthrough experiments and deduced competitive CO2–H2O loadings... | Download Scientific Diagram

Data-Driven Many-Body Models for Molecular Fluids: CO2/H2O Mixtures as a Case Study | Journal of Chemical Theory and Computation

SOLVED: Balance the following equation and label the type of reaction taking place: C5H9O + O2 â†' CO2 + H2O