
CuO + H2CO3 –––> CuCO3 + H2O. Beautiful copper II carbonate ❤ | Teaching chemistry, Chemistry education, Science chemistry

Poudre de bleu-vert d'alimentation de l'usine Cuco3 de base de carbonate de cuivre - Chine Poudre de carbonate de cuivre, de la poudre de carbonate cuivrique

Example: CuCO3(s) + H2SO4(aq) CuSO4(aq) CO2(g) + H2O Add the excess solid to the acid until no more reacts. Heating may be required Filter off the excess. - ppt download
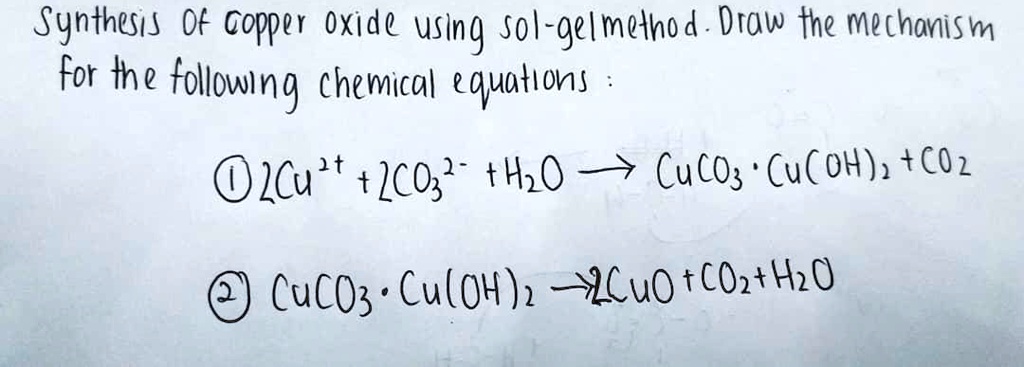
SOLVED: Synthesis of Copper Oxide using Sol-gel method. Draw the mechanism for the following chemical equation: Cu(OH)2 + 2CO3 -> CuCO3 + H2O. CuCO3 -> Cu(OH)2 + CO2. CuCO3 -> CuO + CO2 + H2O.

Copper Carbonate,Senior Chemistry - Extended Experimental In-Industry News-Nickel Acetate,Cobalt Sulfate-Fairsky Industrial Co., Limited
Solved: Copper carbonate (CuCO_3) reacts with hydrochloric acid (HCl) according to this equation: [algebra]
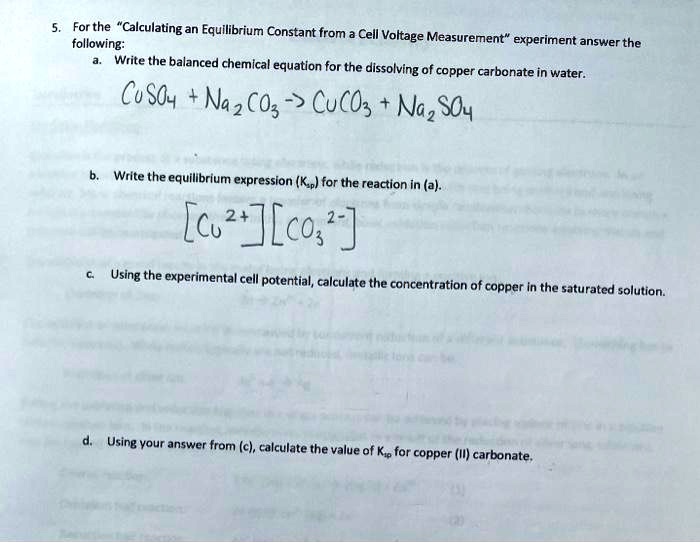








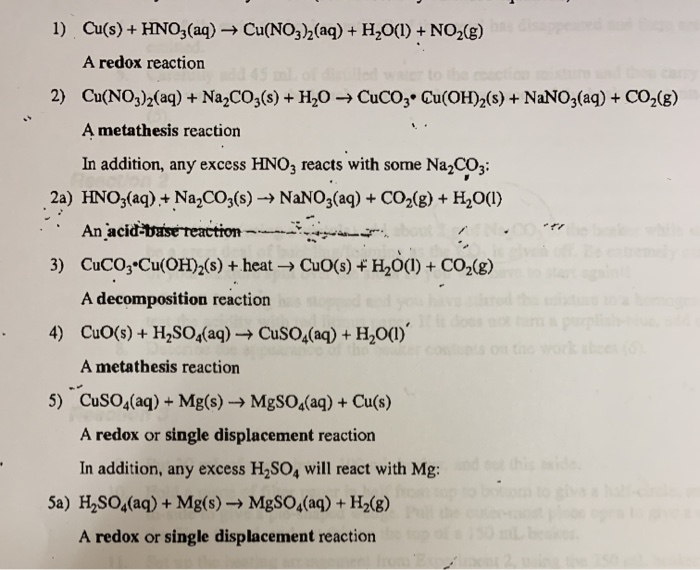


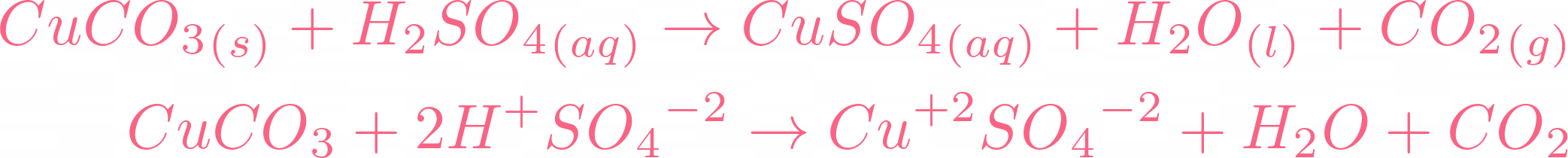



%20carbonate%20basic%20,%20CuCO3.CU(OH)2.H2O,%20%C4%90%C3%B4%CC%80ng(II)cacbonat%20baz%C6%A1%20,%20S%C6%A0N%20%C4%90%E1%BA%A6U%20,%20JHD%20CHEMICAL%20-%20Copy.jpg)